 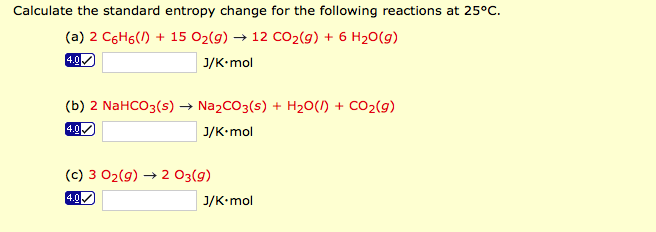
This simulation models this in an extremely simple way by modifying the ‘Entropy and disorder’ simulation so that the intrinsic rates of forward and back reactions (ie the probability of A converting to B or B converting to A) can be varied. Under certain circumstances, this may result in a situation where the amounts of reactants and products remain the same (‘an equilibrium mixture’) even though both forward and back reactions are still continuing. The approach to equilibriumĪ reversible reaction has two reactions, forward and back occurring at the same time. Between these temperatures, the reaction is considered to be reversible and in a closed system an equilibrium would be set up containing some of all the reactants and products concerned. If the reaction were to take place in a closed system, an equilibrium would be set up.Īs a rule of thumb, reactions with Δ G more negative than –60 kJ mol -1 are considered to go to completion while those with Δ G more positive than +60 kJ mol -1 are considered not to occur at all. In fact, the reaction does not simply ‘flip’ between feasibility and non-feasibility (or going in one direction or the other). 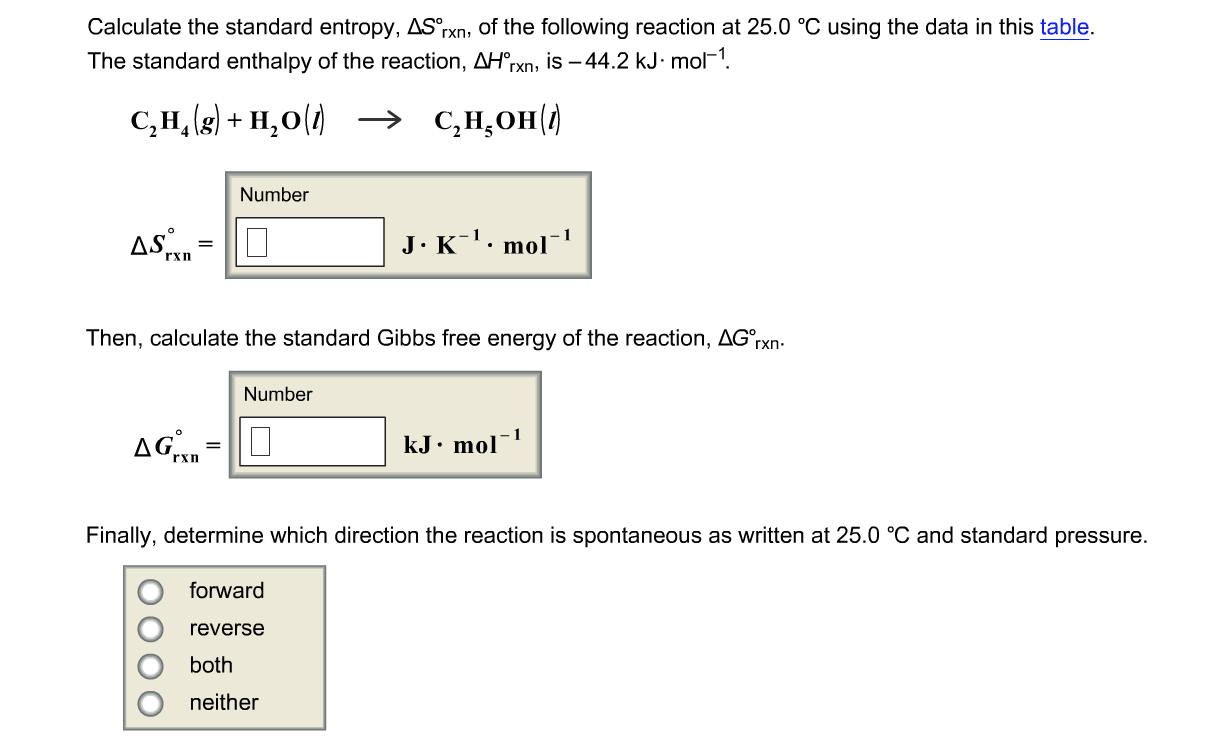
Δ S system may change with temperature. For example, if one of the reactants or products changes state (melts or boils) this will affect Δ S system. This is an approximation only, but for most reactions the change with temperature is small. Throughout these calculations, we have assumed that Δ H does not change with temperature. This explains why some reactions go in one direction at one temperature and in the opposite direction at a different temperature.įor example, let us look at the reaction between calcium oxide and carbon dioxide (considered in the tutorial on chemical reactions and direction):įor this reaction, Δ H is –178 kJ mol -1 (ie –178 000 J mol -1) and Δ S system is –161 J K -1 mol -1. Sometimes, changing the temperature can change the sign of Δ G. We can see from the expression for the Gibbs free energy that the value of Δ G depends on temperature: The temperature dependence of the Gibbs free energy It is also an energy term, which is a concept more familiar to most chemists than entropy. This is what makes this quantity so useful to chemists. Notice also that all the terms in the expression relate to the system rather than the surroundings. Notice that if Δ G is negative, the reaction is feasible. We call the term ‘– TΔ S total’ the Gibbs free energy, after the American chemist Josiah Willard Gibbs. So it is necessary to convert the units, usually by dividing the entropy values by 1000 so that they are measured in kJ K -1 mol -1. 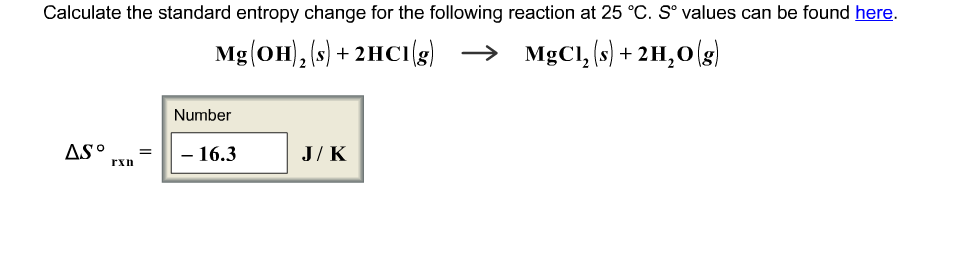
Chemists normally measure energy (both enthalpy and Gibbs free energy) in kJ mol -1 (kilojoules per mole) but measure entropy in J K -1 mol -1 (joules per kelvin per mole). We must take care when using mathematical expressions that include both energy and entropy. RSC Yusuf Hamied Inspirational Science Programme.Introductory maths for higher education.The physics of restoration and conservation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
